Naval Facilities (NAVFAC) Engineering and Expeditionary Warfare Center (EXWC) has responded to the COVID-19 pandemic through the use of the command’s additive manufacturing capabilities.
NAVFAC EXWC will soon deliver the first batch of 250 face mask kits to commands needing additional PPE. Other commands will continue to coordinate with NAVFAC EXWC leadership for shipment of needed PPE, and subsequently update the additive manufacturing teams on further developing supplies.
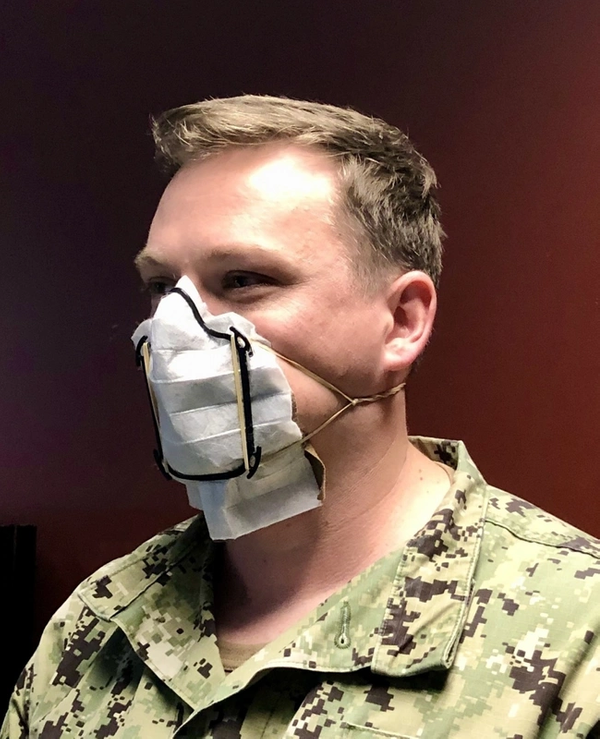
The face masks kits are being printed in three sizes: small, medium, and large. In addition, the design of the face mask frame can be heat molded to conform to the end user’s face using a low-level heat gun or blow dryer.
Producing a single face mask takes about 20 minutes, including setup and cleaning. Each mask’s kit includes an American-made MERV13 filter that provide some viral filtration, a 3-D printed frame, four rubber bands, and a cloth patch — all disinfected and carefully packaged in a plastic bag.
Between three to six at a time are printed per printer tray. At full capacity, the printers can produce over 50 frames per hour. Seven-person production crews from NAVFAC EXWC and NCG-1 are simultaneously tending to printers, breaking down filters, cutting patches, sanitizing mask frames, packaging face mask kits, and providing quality control support.
The process began in late March, when NAVFAC EXWC’s dry lab and the Expeditionary Maintenance Center (EMC) rapidly began developing a workflow at the request of the Assistant Secretary of the Navy for Research, Development & Acquisition (RD&A). Tactical efforts began April 9 at the direction of NAVFAC EXWCAdditive Manufacturing Implementation Lead Todd Jonas.
“By nature, engineers live to figure out solutions,” said Jonas, a certified professional engineer. “Our team of NAVFAC and Naval Construction Group ONE (NCG-1) professionals collaborated and executed this rapid response for 3-D printing of face masks for urgent fleet needs.
"This endeavor is focused solely on preparing for a response if called upon. Being ready now, our efforts will allow NAVFAC to accelerate the production and shipment of needed personal protective equipment.”
On April 7, Naval Air Systems Command (NAVAIR) released a technical data package to the U.S. military community. It provided a sample face mask kit validated by the National Institutes of Health and Department of Veterans Affairs, and approved for emergency use by the Food and Drug Administration (FDA) when social distancing is not a viable option.
Once authorized by leadership, NAVFAC EXWC and NCG-1 developed several “proof of concept” face mask kits using NAVAIR’s technical data package. NAVFAC EXWC and NCG-1 then coordinated the feasibility, cost, and procurement of the face mask kits for the United States Naval Ship (USNS) Mercy, currently moored at the Port of Los Angeles.
“Our team jumped at the chance to leverage the capability we’ve developed in additive manufacturing for the greater good of the Fleet and the people of the greater Los Angeles area," said NAVFAC EXWC Commanding Officer Capt. Michael Saum. "Being able to partner with the Pacific Fleet Seabees makes it even more rewarding.
“This global COVID-19 pandemic does not deter [NAVFAC] EXWC from our mission to provide specialized expeditionary solutions to the warfighter," Saum continued. "The fact that we continue to support the warfighter, alongside the warfighter, and meet the needs of our community, gives our team a great sense of purpose.”
The additive manufacturing teams from EXWC and NCG-1 began coordinating a response to various requests from Jonas, who received orders from NAVFAC headquarters. Teams began updating their additive manufacturing equipment capability to produce requested parts and then determined how many parts could be produced within 24 hours. Teams then coordinated their workforce availability to support production, and generated workflows to ensure the safe shipment of the face mask kits.
The additive manufacturing capabilities of NAVFAC EXWC offer both CONUS and OCONUS support options that are legally produced and vetted by the FDA. As situations continue to develop, NAVFAC EXWC is positioned to lean forward to tactically and strategically support the COVID-19 support efforts as requested.



